Recommended as first-line treatment per the
RRP Foundation position
statement.1,2
The results are CLEAR
Select study design
PAPZIMEOS was evaluated in an open-label, single-arm study of 38 adult patients with recurrent respiratory papillomatosis (RRP) in the United States. 35/38 patients received the recommended dosage of 5×1011 particle units (PUs) per injection.1 See full study design.
51% of patients (n=18/35) did not require any surgeries in the year following PAPZIMEOS treatment1
Complete responses have been durable and are ongoing1
| 1 YEAR |
Primary endpoint: complete response 51% OF COMPLETE (n=18/35) did not require any surgical interventions for 1 year post treatment (95% CI: 34%-69%).1 |
|
| 2 YEARS |
Durability of complete response 15 out of 18 complete responders evaluated at 2 years demonstrated continued complete response.1 |
|
| 3 YEARS |
Complete responses are ongoing, with most remaining durable at a median follow-up of 3 years3 Over a 36-month median follow-up period (range, 27-37 months), a majority of patients (n=15/18) have maintained a complete response. No new safety events were observed during this long-term follow-up.3* |
|
| 4 YEARS |
Ongoing follow-up continues4 |
|
| 5 YEARS |
Planned follow-up completion4 |
The majority of patients had a reduction in surgical interventions in the year post treatment5

106 FEWER SURGERIES OCCURRED
across the entire treatment population in the 1 year following treatment vs the year before treatment.5
In the year prior to treatment, patients had a median of 4 surgeries (range, 3-10).5
Data do not include 1 patient who did not complete the 12-month follow-up and was considered a nonresponder.5
Select Important Safety Information
WARNINGS AND PRECAUTIONS
Injection-Site Reactions: Injection-site reactions have occurred with PAPZIMEOS injection. Monitor patients for local site reactions for at least 30 minutes after the initial treatment.
Thrombotic Events: Thrombotic events may occur following administration of adenoviral vector-based therapies. Monitor patients for signs and symptoms of thrombotic events and treat events according to clinical practice.
A CLEAR focus on the root cause of RRP
PAPZIMEOS uses the power of the patient’s own immune system to activate a T-cell response to target HPV 6 and 11 in clinical responders1,4
Clinical responses were correlated with T-cell activity in the blood4§
As an immunotherapy, PAPZIMEOS elicits an immune response to help patients clear the chronic infection. PAPZIMEOS treats the underlying condition by eliciting a T-cell response against human papillomavirus (HPV) 6-and HPV 11-infected cells.1,4,6
HPV 6- and HPV 11-specific T-cell responses in patients who demonstrated a clinical response to treatment1,5§
Exploratory endpoint
T-cell responses were
165x higher
than pre-treatment levels1,4
T-cell responses at treatment completion are expressed as a mean fold change from baseline in patients who had a clinical response to treatment (n=19). Nonresponders (n=9) had 5.1x greater T-cell activity at treatment completion compared to pre-treatment levels.4
These data demonstrate that the clinical response of PAPZIMEOS is correlated with an induction of HPV 6- and HPV 11-specific T-cell responses.4
Clinical response is defined as an elimination (complete response [CR]) or ≥50% reduction of surgical debulking procedures (partial response [PR]) in the 12 months following treatment completion.1,4
Select Important Safety Information
USE IN SPECIFIC POPULATIONS
Pregnancy: There are no available data with PAPZIMEOS in pregnant women.
Lactation: There is no information available on the presence of PAPZIMEOS in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for PAPZIMEOS and any potential adverse effects on the breastfed child from PAPZIMEOS or from the underlying maternal condition.
Pediatric Use: The safety and effectiveness of PAPZIMEOS have not been established in pediatric patients.
Geriatric Use: Clinical studies of PAPZIMEOS did not include sufficient numbers of patients 65 years of age and older to determine whether they respond differently from younger patients.
Representative images of complete responders


See a CLEAR difference7||
These 4 patients have shown a continued response beyond 35 months. Patients will continue to be followed for a period of 5 years.3‑5¶
Patient 5
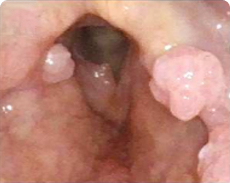
Before PAPZIMEOS treatment

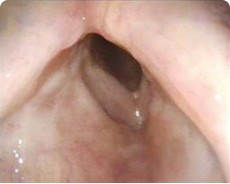
12 months after PAPZIMEOS treatment
Patient 7
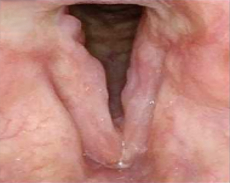
Before PAPZIMEOS treatment

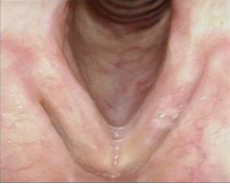
12 months after PAPZIMEOS treatment
Patient 11
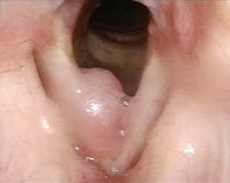
Before PAPZIMEOS treatment

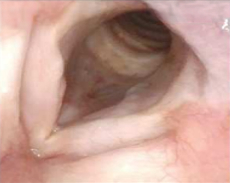
12 months after PAPZIMEOS treatment
Patient 13
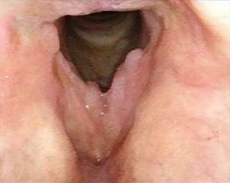
Before PAPZIMEOS treatment

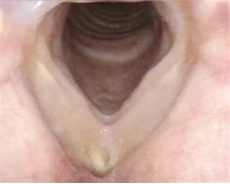
12 months after PAPZIMEOS treatment
51% (n=18/35) of patients were complete responders. Laryngoscopic results are displayed for 4 of the 18 complete responders. Post-treatment images were obtained 12 months after completion of study treatment. These do not represent all patients treated with PAPZIMEOS. Results may vary.1,7
As of September 19, 2025.3
PAPZIMEOS was studied in an open‑label, single‑arm, phase 1/2 trial in adult patients with RRP1,3‑5

KEY ELIGIBILITY CRITERIA3:
- Adults aged 18 years or older with a clinical diagnosis of RRP
- Required ≥3 surgical interventions in the 12 months before treatment
- Eastern Cooperative Oncology Group Performance Status of 0 or 1
SQ=subcutaneous.
In the phase 1 portion of the study, 2 dose levels were tested: 1 x 1011 and 5 x 1011 PU. A total of 38 patients were enrolled in the trial. The efficacy population includes 35 of the 38 patients who were treated at a dose of 5 x 1011 PU per injection.3
All patients underwent a surgical debulking procedure before the first administration of PAPZIMEOS.3
If patients had visible regrowth of papillomas during the treatment interval, a surgical intervention could be performed prior to the third and fourth dose to maintain minimal residual disease.3
Clinical endpoints
PRIMARY ENDPOINT
- Complete response (CR) rate: Percent of patients with no surgical interventions during the 12
months after treatment3
KEY SECONDARY ENDPOINTS
- Partial response (PR) rate: Percent of patients with at least a 50% decrease in the number of interventions in the 12 months post treatment compared to the 12
months pre treatment3 - Objective response rate (ORR): Percent of patients with a complete or partial response3
- Safety evaluation3
EXPLORATORY ENDPOINTS
- T-cell response4
